Drug price changes hit the deep freeze after the early 2026 flurry
As a non-profit, we at 46brooklyn have made it our goal to provide insights into U.S. drug pricing data available in the public domain based upon the figures we’ve gathered over the prior month. This month we review the happenings of drug pricing in our shortest month – February. But before we get into the details, first, let’s take a look at some of the latest news hitting the drug supply chain:
Drug Pricing News
As we move further into 2026, there are going to be several big-name medications that will be losing their exclusivity, which allows for competition to enter the market and potentially lower drug costs for patients and plan sponsors (as long as the drug channel passes along those savings, of course). Therapeutic areas such as diabetes have been hot drug classes with blockbuster medications like GLP-1s, while other diabetes medication classes have essentially become less popular mainly due to decreased efficacy compared to newer medicines within the therapy realm.
Januvia and Janumet are set to lose their patent exclusivity in 2026. Januvia (sitagliptin) was notably the 1st DPP-4 inhibitor that was approved 20 years ago in October 2006. Sitagliptan is also available in an authorized generic formulation known as Zituvio, which was approved in 2023. An authorized generic is the same exact ingredients (even the inactive) as the brand name medication. Similarly, another DPP-4, saxagliptan, was one of the generic medications reported on as an outlier this month from our NADAC Change Packed Bubble Chart that also decreased in price.
A few of the medications identified to lose patents in an article from Fierce Pharma included Ibrance and Eliquis, which have U.S. patent protections that end in 2027. Our data shows that in February, Ibrance’s price decreased while the price for Eliquis increased. It can be difficult to determine the sentiment behind why a medication price increases or decreases, but if there were obvious answers, you wouldn’t be on this website, that’s for sure.
The Drug Pricing Cliff Notes
After a massive wave of drug price changes in January, we have hit the boring lull of February, where there were a net (combined increases minus decreases) of 30 brand drug list price increases in February, with price change percentages ranging from a decrease of -70.0% (Humalog KwikPen & LYUMJEV) to an increase of 35.5% (OMISIRGE).
Typically, we see price changes all over the board, some of which can mean an impact of just a few hundred dollars, whereas others are hundreds of millions of dollars or in some rare instances, billions of dollars. In February, some medications have as low as a -70.0% price decrease whereas others have an increase of up to 35.5%, which is much lower than we saw at the beginning of the year. However, depending on the price of a medication, even a small overall percentage increase could be substantial enough to be thousands or even millions of dollars compounded over time.
In February, six medications took a price decrease, which is a decrease compared to last month where we saw 20. The six medications decreasing in price is a steady amount that we typically see when it comes to medications decreasing in cost. While WAC price decreases are generally considered a good thing, as we know, these decreases can affect drugmaker rebates. Therefore, if the WAC of a brand medication goes down, this also can impact the amount of rebates and discounts that drugmakers offer on that drug (if this is a foreign concept to you, we recommend you learn more about drugmaker rebates and the “gross-to-net bubble” of brand-name medications.
Some of the biggest and/or most interesting movers to take note of for February 2026 were:
o Injectafer Solution for injection (5.0% increase; $34 million gross Prior Year Medicaid Expenditures [PYME])
o Humalog KwikPen Solution for injection, Prefilled Pen (-70.0% decrease; $64 million PYME)
o Gvoke HypoPen Solution for injection (3.0% increase; $35 million PYME)
o Brixadi Solution for injection, Extended Release (4.0% increase; $46 million PYME)
o Tremfya Solution for injection (5.0% increase; $366 million PYME)
o Xarelto Oral tablet (2.3% increase; $455 million PYME)
o Ibrance Oral capsule (-17.9% decrease; $22 million PYME)
o Fanapt Oral tablet (4.0% increase; $44 million PYME)
Some standout price increases are for medical conditions including low blood sugar (Gvoke HypoPen), low iron (Injectafer Solution), opioid use disorder (Brixadi), autoimmune disorder (Tremfya), and mental health (Fanapt).
Some standout price decreases are for diabetes (Humalog KwikPen) and breast cancer (Ibrance).
Although the percentage increase wasn’t high (5.0%), the dollar amount for Tremfya impacted $366 million in PYME. Therefore, it is important to note that sometimes a similar or even a smaller percentage increase (especially for an already expensive brand medication) can result in a significant chunk of change in in overall gross drug spending. As mentioned above, bear in mind that as you read those numbers, that they are the prices before drugmaker rebates, which as we know are growing significantly over time and are at their largest amounts in the Medicaid and 340B programs.
On the generic side of the coin, year-over-year (YoY) generic oral solid price changes are at -13.4%, but you probably won’t see much of that in the headline drug pricing news (although we note at least some coverage over oil prices potentially increasing generic drug prices in the short-term). The facts are, most of us take generic drugs; so if we’re playing the odds, the issues impacting generic drugs are potentially more individually important than the brands (but brands get all the headlines because they cost so much more than the generics).
And that is the Cliff Notes. If you want to find out how we arrived at these insights, read on. Otherwise, check back with us next month for the March numbers.
What we saw from brand-name medications in February
1. A relatively stable and historically aligned number of list price increases for brand drugs
There were a total of 30 brand-name medications that saw wholesale acquisition cost (WAC) price increases in February, which is featured and contextualized in our Brand Drug List Price Change Box Score. This is a similar number of increases compared to what we have seen with the month of February in years’ past (at least for the last few years) with around 30-50 brand name drug price increases.
Price changes this month ranged from -70.0% to 35.5%.
As a reminder, brand price increases in Medicaid are largely held in check thanks to the Medicaid Drug Rebate Program (MDRP), which includes rebate penalties for drug price increases that occur faster then the rate of inflation. Medicare now has similar cost containment provisions. Commercial employers and cash-paying patients may lack protections from price increases – especially those that occur faster than the rate of inflation.
This is one of a number of reasons that solely analyzing brand list price changes provides an incomplete picture of what’s really happening with brand manufacturer economics, thanks to the growing lot of opaque rebates, discounts, and giveaways that drugmakers shave off those list prices. But alas, until drugmakers, PBMs, insurers, wholesalers, 340B covered entities, and rebate aggregators make more granular data on net prices public, we’ll continue working with what we’ve got.
2. Brand price trends over time
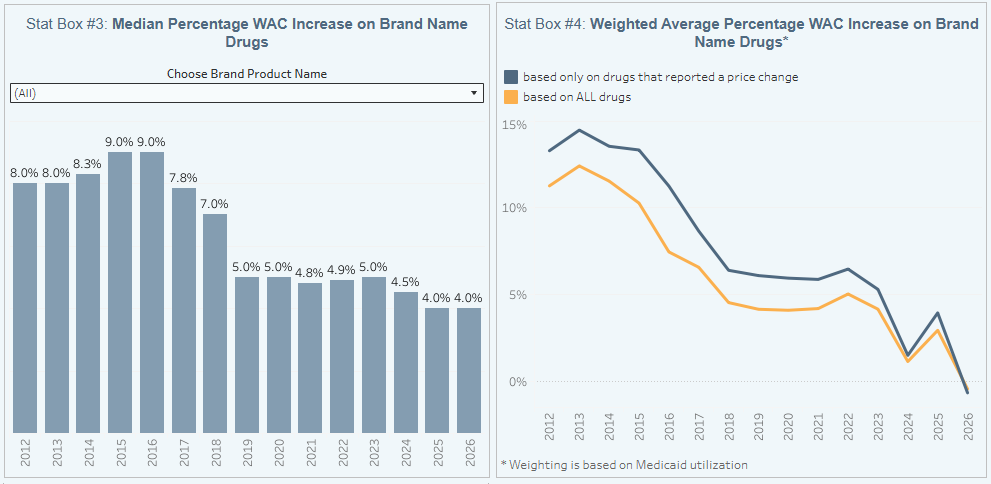
To help contextualize brand name drug list price increase behavior, we find it beneficial to review past trends. In comparison to the data from prior months of February, this year seems to be decently close with February 2021, which also had 30 (combined increase and decreases) branded list price changes. Looking at past trends, February is ALWAYS a month where there have consistently been a small number of branded price changes, with the total this February being in line with the last few years’ worth of data.
Figure 1
Source: Elsevier Gold Standard Drug Database, CMS State Drug Utilization Data, 46brooklyn Research
The highest number occurred number years ago in February 2017 with 63 net branded price increases, whereas the lowest was in February of 2020, when there were only 14 price increases.
To put it into a more recent perspective (over the last five years), in February 2024, there were a net (combined increases and decreases) of 36 increases, 53 in February 2023, 41 in February 2022, 30 for February 2021, and 14 in February 2020.
Moreover, when further examining our brand drug box score visualization, we continue to see February being the time of year where there is a very small amount of change for brand price increases with a also a low number of brand price decreases.
Of the drugs that took increases so far this year, the median price increase has been 4.0% — a percentage that has not fluctuated much since 2019.
3. Brand drug list price changes worth taking note of in February
We identify drugs worth taking note of in a couple different ways. Primarily, we look for medications with a lot of prior Medicaid expenditures (not that Medicaid is the end-all-be-all, but it is the only program that regularly publishes past utilization with some decent granularity). We next look for drugs with large pricing changes (+/- 10%). And finally, we look for drugs that are interesting for us either because we’ve previously written on them or because we find them of unique clinical value. And given the number of price changes for brand name drugs in January, well we have a lot to potentially talk about.
Humalog KwikPen solution for injection (insulin lispro) is a rapid acting human insulin analog indicated to improve glycemic control in adult and pediatric patients with diabetes mellitus.
This medication took a decrease in WAC of -70.0% resulting in $34 million in gross prior year Medicaid expenditures (PYME).
An announcement for Humalog KwikPen as of February 1, 2026, indicated that there would be a 70% reduction in WAC, which would affect the U-200 product and was spot on for what we saw in our data this month as well.
When it comes to lowering the cost of insulin products, Lilly also has their Lilly Insulin Value Program that provides all Lilly insulins to a patient for $35 a month for those with commercial or no insurance. Insulin affordability has been a hot topic for several years now sparking a lot of debate around election time. Insulin pricing caps have been put in place by various states as well as government funding insurance programs, Medicaid, and Medicare. The Inflation Reduction Act (IRA) of 2022 makes sure that Medicaid and Medicare recipients pay no more than $35 per month. Many manufacturers of insulin also created additional programs for patients with commercial insurance or no insurance at all (i.e. Lilly, Novo Nordisk, Sanofi, Mannkind). So, it does not seem to come as a surprise that the cost of this medication has decreased due to the recent years’ insulin spotlight and the direct call out by Lilly.
Gvoke HypoPen solution for injection (glucagon injection) is an antihypoglycemic agent indicated for subcutaneous use for the treatment of severe hypoglycemia in adult and pediatric patients aged 2 years and older with diabetes.
This medication saw an increase in WAC of 3.0%, which resulted in an additional $35 million in gross prior year Medicaid expenditures (PYME).
Gvoke HypoPen was the 1st ready-to-use liquid glucagon available in a single dose vial and syringe kit, which eliminated the need for the medication to be reconstituted a rate-limiting step during an emergency situation of low blood sugar. This product was approved in 2021 and started distribution in early Q1 of 2022. In March 2025, Gvoke VialDx received FDA approval to use the same medication as an intravenous diagnostic aid to temporarily stop the movement of the gastrointestinal tract in adult patients, which launched not long ago in August 2025.
Injectafer solution for injection (ferric carboxymaltose) is an iron replacement product indicated for the treatment of:
iron deficiency anemia (IDA) in:
adult and pediatric patients 1 year of age and older who have either intolerance or an unsatisfactory response to oral iron.
adult patients who have non-dialysis dependent chronic kidney disease.
iron deficiency in adult patients with heart failure and New York Heart Association class II/III to improve exercise capacity.
This medication experienced an increase in WAC of 5.0%, or in other words, $34 million in gross prior year Medicaid expenditures (PYME).
Injectafer is not new to price increases, as we reported in 2021 on this medication with a WAC increase of 3%, which impacted $33.8 million in gross PYME at the time and is almost identical to what we saw in February 2026 as well.
Tremfya Solution for injection (guselkumab) is an interleukin-23 antagonist indicated for the treatment of:
Adults and pediatric patients 6 years of age and older who also weigh at least 40 kg with moderate-to-severe plaque psoriasis and who are candidates for systemic therapy or phototherapy.
Adults and pediatric patients 6 years of age and older who also weigh at least 40 kg with active psoriatic arthritis.
Adults with moderately to severely active ulcerative colitis.
Adults with moderately to severely active Crohn’s disease.
This medication had an increase in WAC of 5.0%, resulting in an increase of $366 million in gross prior year Medicaid expenditures (PYME).
A lot has been going on for Tremfya towards the end of 2025 and into 2026. In September 2025, Tremfya received FDA approval for pediatric patients 6 years and older who weigh at least 40 kg (88 lbs) with moderate to severe plaque psoriasis. Tremfya has become the first and only IL-23 inhibitor (think Skyrizi, Ilumya, Stelara’s of the world) approved for these pediatric indications.
Also in September 2025, Tremfya launched a full subcutaneous regimen that allows self-administration from the start of treatment for the induction dosing phase in patients with Crohn’s disease. This once again makes Tremfya the 1st IL-23 inhibitor to offer both subcutaneous and intravenous induction options for both ulcerative colitis and Crohn’s disease.
And if that wasn’t all … on February 21, 2026, J&J reported results from their long-term extension study, QUASAR, showing that Tremfya sustained clinical, endoscopic, and histologic outcomes through week 140 in adults with moderately to severely active ulcerative colitis. Fierce Pharma is also reporting that Tremfya has been spending a lot of cash on TV ads for both 2025 and as we continue to progress into 2026.
Fanapt oral tablet (iloperidone) is an atypical antipsychotic indicated for:
Treatment of schizophrenia in adults.
Acute treatment of manic or mixed episodes associated with bipolar I disorder in adults.
This medication experienced an increase in WAC by 4.0%, resulting in an increase of $44 million in gross prior year Medicaid expenditures (PYME).
Although there is not any news for Fanapt directly, the manufacturer, Vanda Pharmaceuticals, has found a new use for the same/similar drug molecule — milsaperidone (brand name Bysanti), which converts to iloperidone. This medication has the same indication as Fanapt, which is for the acute treatment of manic or mixed episodes associated with bipolar I disorder and for the treatment of schizophrenia in adults.
Ibrance Oral capsule (palbociclib) is a kinase inhibitor indicated:
For the treatment of adult patients with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced or metastatic breast cancer in combination with:
An aromatase inhibitor as initial endocrine-based therapy; or
Fulvestrant in patients with disease progression following endocrine therapy.
In combination with inavolisib and fulvestrant for the treatment of adult patients with endocrine-resistance PIK3CA-mutated, HR-positive, HER2-negative, locally advanced or metastatic breast cancer, as detected by an FDA-approved test, following recurrence on or after completing adjuvant endocrine therapy.
Notably, this medication had a -17.9% decrease in WAC, resulting in a $22 million increase in gross prior year Medicaid expenditures (PYME). It’s a bit odd that Ibrance is showing up with a price decrease, as this medication was listed by Pfizer as one of their 80 medications that was going to go up in 2026. So maybe there was a Valentine’s Day change of heart? How sweet.
Xarelto oral tablet (rivaroxaban) is a factor Xa inhibitor indicated:
to reduce risk of stroke and systemic embolism in nonvalvular atrial fibrillation
for treatment of deep vein thrombosis (DVT)
for treatment of pulmonary embolism (PE)
for reduction in the risk of recurrence of DVT or PE
for the prophylaxis of DVT, which may lead to PE in patients undergoing knee or hip replacement surgery
for prophylaxis of venous thromboembolism (VTE) in acutely ill medical patients
to reduce the risk of major cardiovascular events in patients with coronary artery disease (CAD)
to reduce the risk of major thrombotic vascular events in patients with peripheral artery disease (PAD), including patients after recent lower extremity revascularization due to symptomatic PAD
for treatment of VTE and reduction in the risk of recurrent VTE in pediatric patients from birth to less than 18 years
for thromboprophylaxis in pediatric patients 2 years and older with congenital heart disease after the Fontan procedure
This medication took a 2.3% increase in WAC, which is $455 million in gross prior year Medicaid expenditures (PYME).
While the Xarelto list price has gone up, it is a bit conflicting considering it was one of the first 10 drugs selected for the Medicare drug price negotiation program that began on January 1, 2026. Perhaps the manufacturer is trying to recoup some of the pricing concessions it provided to the government through other channels?
Brixadi Solution for injection, Extended Release (buprenorphine) contains buprenorphine, a partial opioid agonist.
Brixadi is indicated for the treatment of moderate to severe opioid use disorder in patients who have initiated treatment with a single dose of a transmucosal buprenorphine product or who are already being treated with buprenorphine.
Brixadi should be used as part of a complete treatment plan that includes counseling and psychosocial support.
This medication took a 4.0% increase in WAC, which is $46 million in gross prior year Medicaid expenditures (PYME).
As of March 16, 2026, the results from a National Institute for Health (NIH) funded study with Brixadi shows that treatment with this injectable medication has greater chances of abstinence from illicit opioids compared to sublingual buprenorphine. The price increase of this medication could be a coincidence, but it is interesting timing given the recent results.
What we saw from generic medications in February
4. A relatively favorable, unweighted price change picture
Each month, we look at how many generic drugs went up and down in the latest month’s survey of retail pharmacy acquisition costs (based on National Average Drug Acquisition Cost, NADAC), and compare that to the prior month (Figure 2).
Basically, the quick way to read Figure 2 is to look for orange bars that are taller than blue bars to the left of the blue line, and exactly the opposite to the right of the line. That would indicate a good month – more generic drugs going down in price compared to the prior month, and less drug prices going up.
Figure 2
Source: Data.Medicaid.gov, 46brooklyn Research
In looking at the above, for every generic drug that increased in price this past month, ~2.9 decreased in price – a favorable view of generic drug price decreases. But as usual, take this unweighted price change analysis with a grain of salt. To really make heads or tails of all of these pricing changes, let’s weight these changes.
5. Weighted Medicaid generic drug costs come in at $58 million in deflation
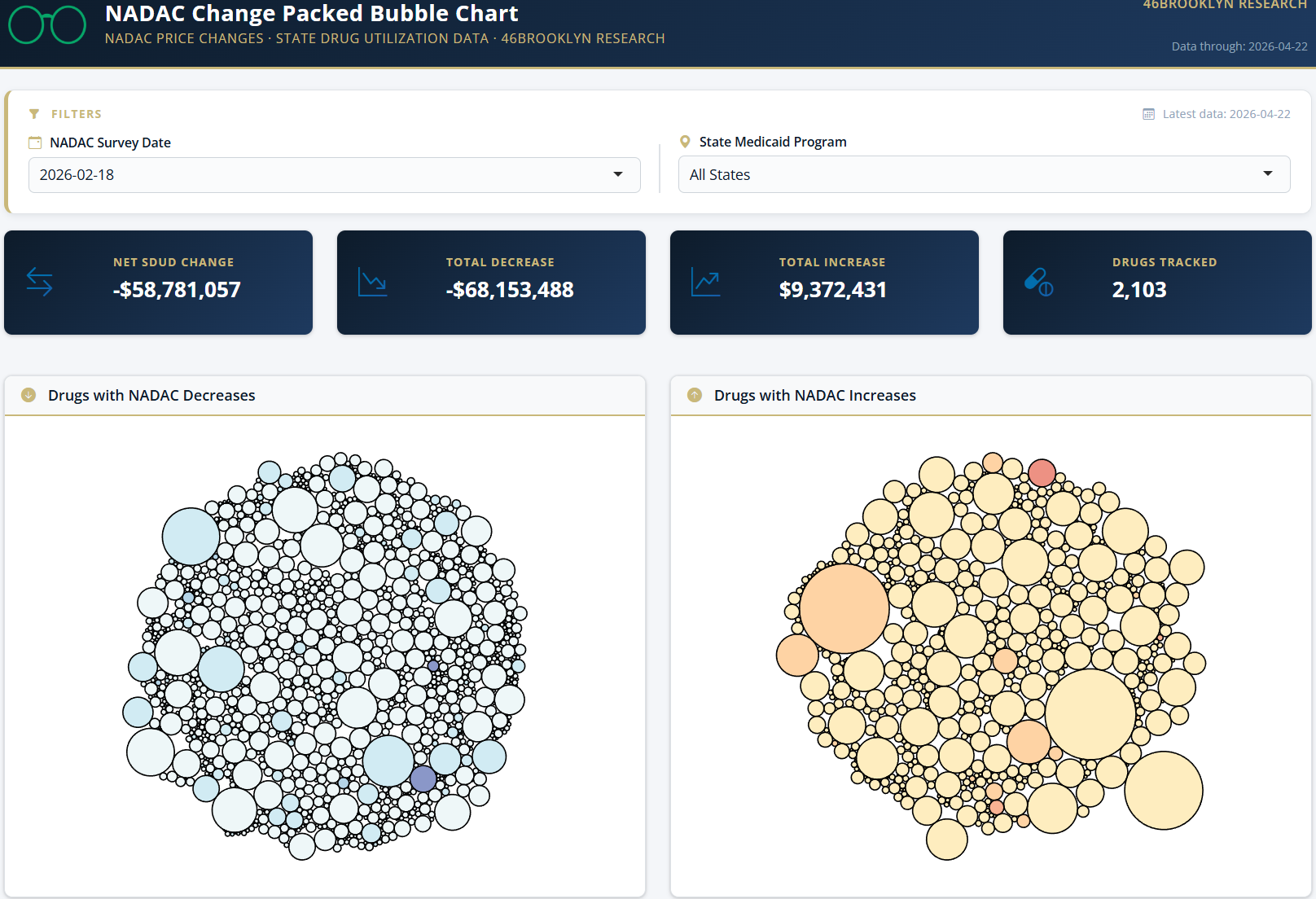
The purpose of our NADAC Change Packed Bubble Chart (Figure 3) is to apply utilization (drug mix) to each month’s NADAC price changes to better assess the impact. We use Medicaid’s 2020 drug mix from CMS to arrive at an estimate of the total dollar impact of the latest NADAC pricing update. This helps quantify what should be the real effect of those price changes from a payer’s perspective (in our case Medicaid; individual results will vary).
The blue bubbles on the left of the Bubble Chart viz (screenshot below in Figure 3) are the generic drugs that experienced a price decline (i.e. got cheaper) in the latest survey, while the yellow/orange/red bubbles on the right are those drugs that experienced a price increase. The size of each bubble represents the dollar impact of the drug on state Medicaid programs, based on utilization of the drugs in the most recent trailing 12-month period (i.e. bigger bubbles represent more spending). Stated differently, we simply multiply the latest NADAC survey price changes by aggregate drug utilization in Medicaid over the past full year, add up all the bubbles, and get the total inflation/deflation impact of the survey changes.
Figure 3
Source: Data.Medicaid.gov, 46brooklyn Research
Overall, in February, there was $9 million worth of inflationary drugs, with an offset of $68 million of deflationary generic drugs, netting out to approximately $58 million of generic drug cost deflation for Medicaid.
These numbers are a bit different than what we saw in January, where there was around $16 million in inflationary costs with $53 million in deflationary costs netting out to approximately $37 million.
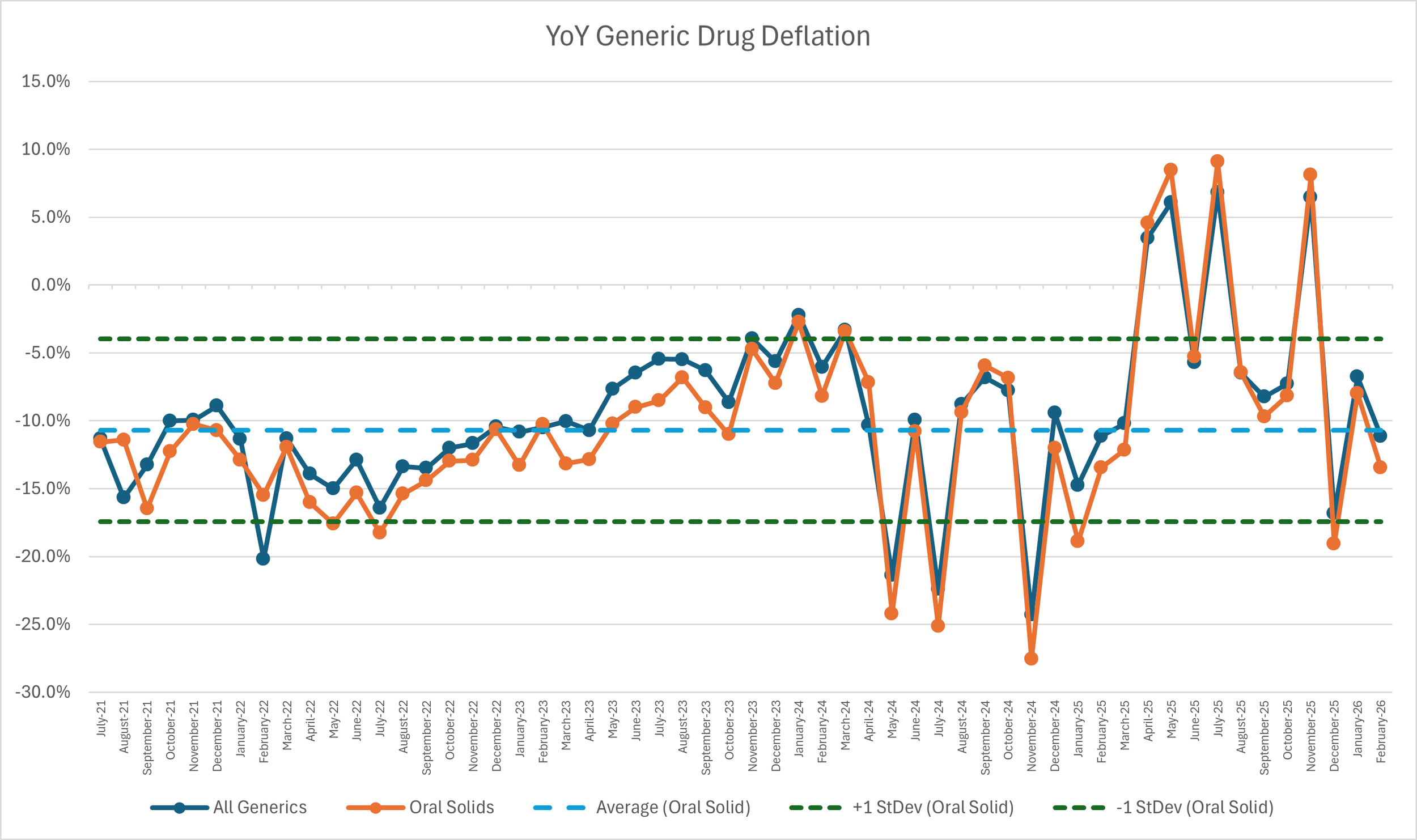
6. Year-over-year generic oral solid deflation at 13.4%
Ever since June 2020, we have been tracking year-over-year (YoY) generic deflation for all generic drugs that have a NADAC price. We once again weight all price changes using Medicaid’s drug utilization data. This past month, deflation on oral solid generics and all generics was at 11% and 9%, respectively (Figure 4). If you are a purchaser of generic drugs, an increase in this metric is ideal as it means costs are declining. This is right in line with the NADAC packed bubble chart, which also showed the cost of generic drugs decreasing. Businesses generally enjoy it when their input costs go down.
Figure 4
Source: Data.Medicaid.gov, 46brooklyn Research
7. Top/notable generic drug decreases this month
For February, the drug price changes total $58 million in deflation based on Medicaid drug utilization. After reviewing the various sizes and colors that comprise the month-over-month changes, there are some notable decreases.
Among the changes, there are a few medications that showed up in the January data and are presenting again as well as some new ones that have popped up this month. It can be typical when you see one strength of a generic medication that other strengths may also be scattered either nearby or throughout the same packed bubble chart. This is an exact scenario that we saw last month in January.
The largest outliers in the packed bubble chart this month include ivabradine 5 mg and 7.5 mg, which decreased in price respectively by -41% and -26%. Last month, these two medications and the same strengths were also considered outliers and showed up with price decreases of -46% for the 5 mg tablet and -21% for the 7.5 mg tablets. New outliers this month include temozolomide 20 mg capsules at -49%, saxagliptan 5 mg tablet, and zileutin ER 600 mg tablet at -14%.
Ivabradine is an oral tablet used to treat heart failure with reduced ejection fraction. Both oral tablet strengths of 5 mg and 7.5 mg took a decrease in price in January by -41% and -26% respectively. Last month when we reported on this medication, there had been analysis of the SHIFT clinical trial from a year ago in March 2025 that showed adding ivabradine to the standard treatment for heart failure significantly improved clinical outcomes, so it continues to draw interest from our perspective that the price continues to decrease even moreso this month despite it being a key medication in treatment.
Temozolomide is a cancer medication used to treat astrocytoma and glioblastoma, which are types of brain or spinal cancers and it decreased by -49%.
Saxagliptan is an oral medication used to treat type 2 diabetes and decreased by -26%. This medication is a DPP-4 inhibitor, which is an older drug class and sadly have fallen out of favor in terms of A1c reduction due to the introduction of newer type 2 diabetes medications such as GLP-1s (i.e. Trulicity, Victoza, Ozempic, etc.). Saxagliptan can be used when additional glucose lowering is needed but GLP-1s are preferred.
8. Top/notable generic drug increases
On the increase side of things, notable this month (just like last month) is that only two of the generic drug increases were darker in color, which means percent changes were on the lower end of the scale — with a couple exceptions. For example, we usually start this section off by noting the most impactful increases of 50% or above, but there aren’t any with that high of a percentage of change.
The two generic medications with the highest increases include tiagabine 12 mg tablet (33% increase) and mepromabate 200 mg tablet (29% increase). Other increases include naproxen DR 375 mg tablet (25% increase) and carvedilol ER 20 mg capsule (20% increase).
Tiagabine 12 mg tablet is an oral medication used to treat focal (partial) seizures and increased by 33%.
Meprobamate 200 mg tablet is an oral medication used to treat anxiety. This medication according to reports has become harder to find in community pharmacies nationwide due to other options being available (i.e. benzodiazepines).
So, are drug prices going up or down? You be the judge. Keep an eye on our dashboards for month-by-month updates. We will see you soon with more fun and depressing drug channel learnings.
Shout-outs to the following publications for featuring our data and insights over the last few weeks:
Will Trump Rx save you money of prescriptions?
WVXU News Cincinnati, 2/26/26
Pharmaceutical giants raise prices on hundreds of drugs despite Trump’s pricing push
New York Sun, 2/24/26
Federal authorities take big steps to rein in drug middlemen at center of Ohio complaints for years
Ohio Capital Journal, 2/18/26
Trump misleads on drug pricing deals
FactCheck.org, 2/18/26
Why are Americans paying more for drugs than anyone else in the world?
WJAG-AM Norfolk, 2/17/26
Feds target PBMs’ hidden fees to benefit consultants
Modern Healthcare, 2/12/26
Money from sick people: How PBMs use AWP, spread, and rebates to inflate drug costs
The Benefits Whisperer, 2/10/26
All about pharmacy benefit managers (PBMs)
Las Vegas Public Affairs Show, 2/7/26
White House debuts drug-buying site TrumpRx, with roughly 40 medications
Wall Street Journal, 2/5/26
White House launches TrumpRx discounted drug site
CBS News, 2/5/26
Drug pricing update
Virginia Focus, 2/5/26
Antonio Ciaccia answers the question, "What's a 'base hit' for the pharmacy profession in 2026?"
Pharmacist’s Voice Podcast, 2/5/26
FTC settles lawsuit with Express Scripts over charges it manipulated insulin prices, impeded access
STAT News, 2/4/26